Biostatistical intepretation series II Clinical Trials Phase I - mitigating misinterpretation risk in dose escalation studies

Adaptive design for identifying maximum tolerated dose early to accelerate dose-finding trial | BMC Medical Research Methodology | Full Text
A Multicenter Phase I/II Study of Obatoclax Mesylate Administered as a 3- or 24-Hour Infusion in Older Patients with Previously Untreated Acute Myeloid Leukemia | PLOS ONE

Innovative design for a phase 1 trial with intra-patient dose escalation: The Crotoxin study. - Abstract - Europe PMC
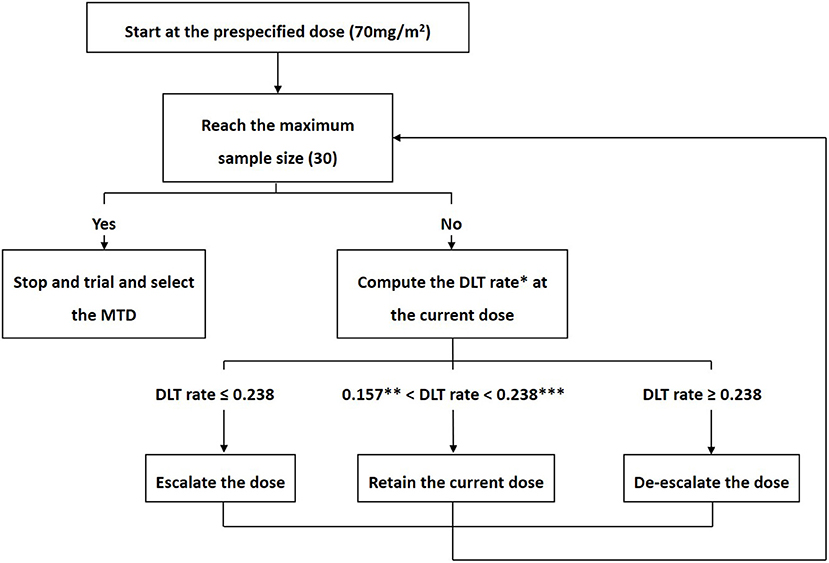
Frontiers | A Dose-Finding Trial for Hyperthermic Intraperitoneal Cisplatin in Gynecological Cancer Patients Receiving Hyperthermic Intraperitoneal Chemotherapy

Bayesian Optimal Interval Design: A Simple and Well-Performing Design for Phase I Oncology Trials. - Abstract - Europe PMC

Study protocol: phase 1 dose escalating study of Pressurized Intra-Peritoneal Aerosol Chemotherapy (PIPAC) with oxaliplatin in peritoneal metastasis

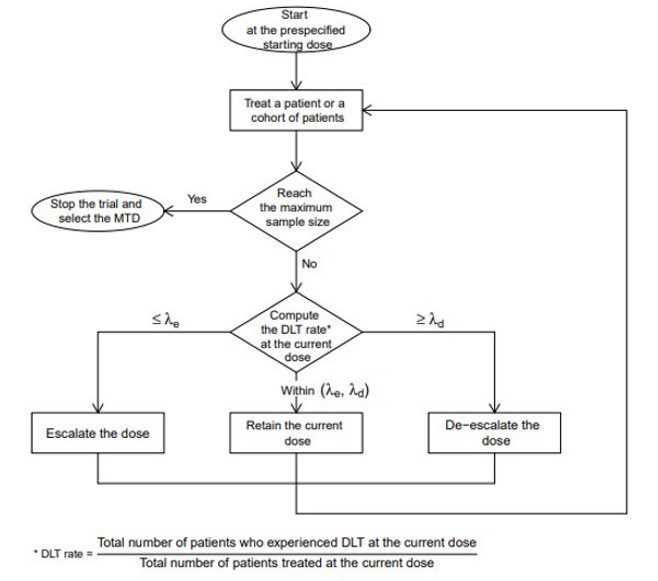
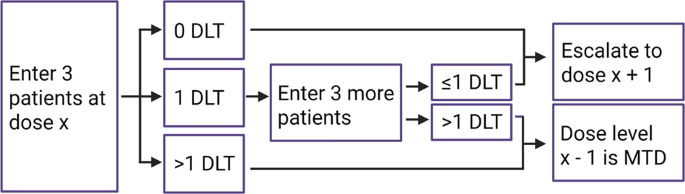
Study design for dose-escalation cohorts and DLT rate (as calculated by... | Download Scientific Diagram

Innovative design for a phase 1 trial with intra-patient dose escalation: The Crotoxin study - ScienceDirect

On Biostatistics and Clinical Trials: Dose Limiting Toxicity (DLT) and Common Toxicity Criteria (CTC) / Common Terminology Criteria for Adverse Events (CTCAE)

Exposure driven dose escalation design with overdose control: Concept and first real life experience in an oncology phase I trial - ScienceDirect